Recently, Beijing Beilu Pharmaceutical Co., Ltd. received the Drug Registration Certificate for its Iopromide Injection, approved and issued by the National Medical Products Administration (NMPA).
The originator of Iopromide Injection is Bayer (Germany), and its indication is for diagnostic use.
Iopromide Injection has been included in the Class A catalog of the National Reimbursable Drug List (NRDL). According to Menet data, across China's three major terminals and six major markets, the sales of Iopromide Injection exceeded RMB 1.6 billion in 2024, and grew by 2.78% to over RMB 400 million in the first quarter of 2025.
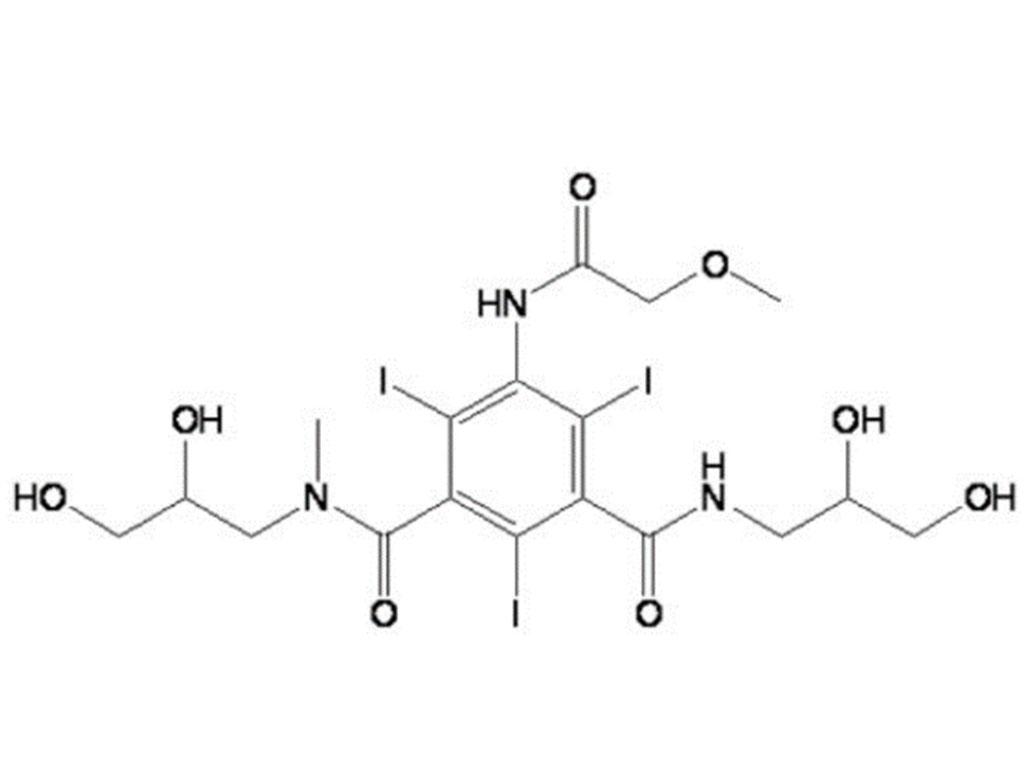
Iopromide Injection was approved as a Class 4 chemical drug, which is deemed to have passed the Generic Drug Quality and Efficacy Consistency Evaluation (GQCE). This approval further enriches the Company's layout of iodinated contrast media products and diversifies its contrast media product line. With the Iopromide Active Pharmaceutical Ingredient (API) of the Company's holding subsidiary, Zhejiang Hichi Pharmaceutical Co., Ltd., approved for marketing in 2025, the "API + Formulation" business model for Iopromide Injection has fully taken shape.
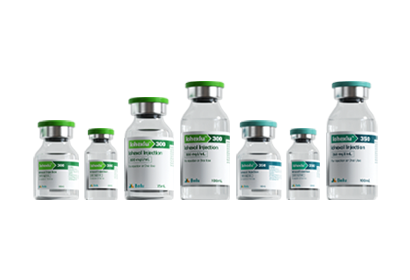
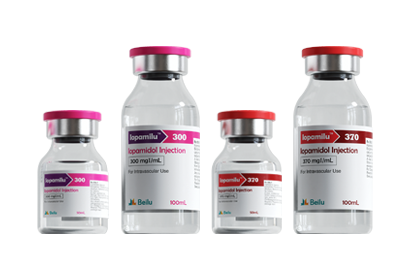
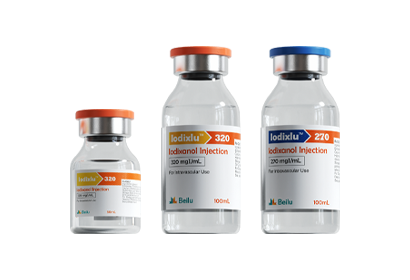
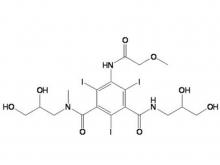
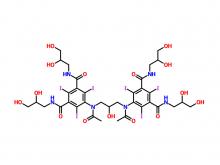
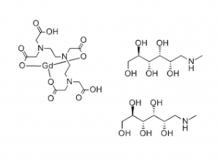
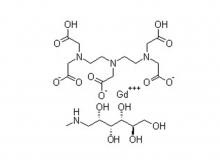
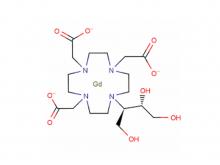
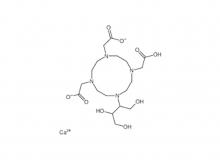
To date, Beilu Pharma has established a comprehensive cluster of iodinated and gadolinium-based contrast media products. Among them, the iodinated contrast media mainly include Iohexol Injection, Iopamidol Injection, Iodixanol Injection, Iomeprol Injection, and the newly approved Iopromide Injection. Regarding gadolinium-based contrast media, Beilu Pharma has rolled out four MRI contrast media: Gadopentetate Dimeglumine Injection, Gadobenate Dimeglumine Injection, Gadobutrol Injection, and Gadoterate Meglumine Injection, covering both macrocyclic and linear gadolinium-based contrast media.
A rich product portfolio helps to better meet diverse clinical medication needs, providing more comprehensive and higher-quality diagnostic solutions for medical institutions at all levels, thereby enhancing the market competitiveness of the Company's products. Beilu Pharma will actively proceed with various preparations for the launch of the new products and aim to bring them to market as soon as possible.
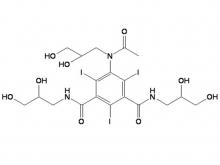
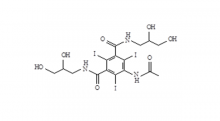 Iohexol Intermediate 5-Amino-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
Iohexol Intermediate 5-Amino-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
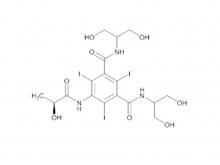
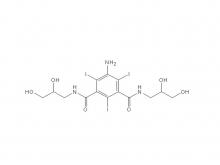 Iohexol/Ioversol Intermediate 5-Amino-N, N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
Iohexol/Ioversol Intermediate 5-Amino-N, N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
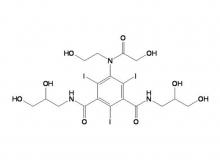
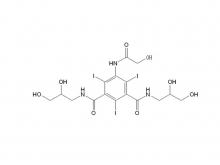 Ioversol Intermediate (order based) N, N'-Bis(2,3-dihydroxypropyl)-5-(glycoloylamino)-2,4,6-triiodoisophthalamide
Ioversol Intermediate (order based) N, N'-Bis(2,3-dihydroxypropyl)-5-(glycoloylamino)-2,4,6-triiodoisophthalamide
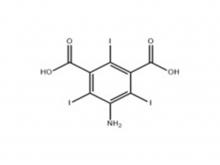 Iopamidol Intermediate (order based) 5-Amino-2,4,6-triiodoisophthalic acid
Iopamidol Intermediate (order based) 5-Amino-2,4,6-triiodoisophthalic acid
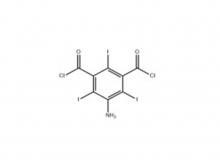 Iopamidol Intermediate (order based) 5-Amino-2,4,6- triiodisophthaloyl acid dichloride
Iopamidol Intermediate (order based) 5-Amino-2,4,6- triiodisophthaloyl acid dichloride
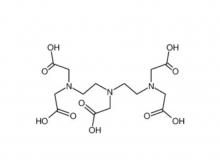 Diethylenetriaminepentaacetic acid (DTPA)
Diethylenetriaminepentaacetic acid (DTPA)

 EN
EN
 jp
jp  fr
fr  de
de  es
es  ru
ru  ar
ar 








 Call us on:
Call us on:  Email Us:
Email Us:  No.3 Shuiyuan West Road, Miyun District, Beijing, China
No.3 Shuiyuan West Road, Miyun District, Beijing, China 