Recently, Beilu Pharmaceutical Jiuwei Zhenxin Granules were successfully selected into the "Science and Innovation China" New Drug Achievement Case Library of the China Association for Science and Technology. This project is one of the outstanding case database construction projects carried out by the China Association for Science and Technology "Science Innovation China" of China’s official platform in various fields...
This project is one of the outstanding case base construction projects carried out by the China Association for Science and Technology "Science and Technology Innovation China" in various fields. In the field of the biomedical industry, 300 cases of excellent new drug achievements will be selected and placed on the official platform of "Science and Technology Innovation China" founded by the China Association for Science and Technology.

The solicitation standard for the case database of this project is that domestically registered biomedical companies including foreign capital, new drugs, improved drugs, and generic drugs (chemical drugs, biological drugs, Chinese medicine, etc.) that have been registered in China since 2016, are used in key technology research and development. Innovations, technological breakthroughs, or unique advantages in clinical efficacy and safety can be selected as exemplary cases.
Jiuwei Zhenxin Granules is the first domestic anti-anxiety Chinese medicine preparation successfully developed by Beilu Pharma. It fills the gap in the treatment of anxiety disorders with Chinese patent medicines. It was approved by the NMPA in 2008 and entered the national medical insurance catalog in 2017. Jiuweizhenxin Granules are derived from the "Pingbu Zhenxin Pill" in the official prescription book "Taiping Huimin Heji Prescription" published in the Song Dynasty. Professor Tuya from Beijing University of Traditional Chinese Medicine combined the knowledge of modern medicine and set the drug screening as 9 flavors.

Jiuwei Zhenxin Granules is the first to carry out pharmacological research on anxiety animal models in China. Phase II and Phase III trials use anti-anxiety drugs as positive controls to carry out multi-center, randomized double-blind clinical studies. In Phase II and Phase III trials, Jiuwei Zhenxin Granules showed significant efficacy, compliance, and good safety, without serious side effects and dependence. After the launch of Jiuwei Zhenxin Granules, large-sample phase IV clinical studies have been actively carried out to further confirm the safety and effectiveness of the combination and individual drugs. Jiuwei Zhenxin Granules can improve the current situation of large adverse reactions of chemical drugs and poor compliance of patients with medication, which is conducive to patients' adherence to medication and disease recovery.
Jiuwei Zhenxin Granules has been recommended by 12 clinical guidelines/industry consensus and national diagnosis and treatment norms/clinical pathways. It is the “Mental Disorders Diagnosis and Treatment Standards (2020 Edition)” issued by the General Office of the National Health Commission. It is in the treatment of generalized anxiety disorder. The only Chinese patent medicine listed.
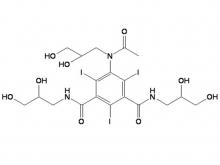
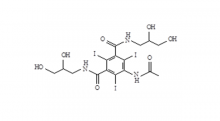 Iohexol Intermediate 5-Amino-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
Iohexol Intermediate 5-Amino-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
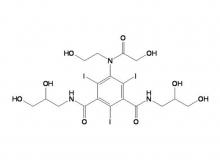
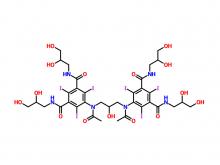
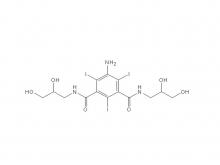 Iohexol/Ioversol Intermediate 5-Amino-N, N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
Iohexol/Ioversol Intermediate 5-Amino-N, N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide
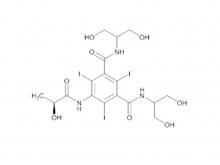
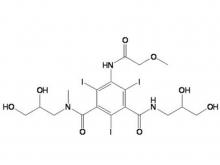
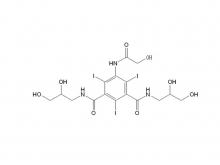 Ioversol Intermediate (order based) N, N'-Bis(2,3-dihydroxypropyl)-5-(glycoloylamino)-2,4,6-triiodoisophthalamide
Ioversol Intermediate (order based) N, N'-Bis(2,3-dihydroxypropyl)-5-(glycoloylamino)-2,4,6-triiodoisophthalamide
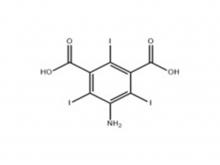 Iopamidol Intermediate (order based) 5-Amino-2,4,6-triiodoisophthalic acid
Iopamidol Intermediate (order based) 5-Amino-2,4,6-triiodoisophthalic acid
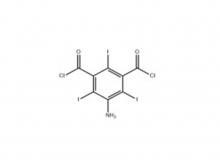 Iopamidol Intermediate (order based) 5-Amino-2,4,6- triiodisophthaloyl acid dichloride
Iopamidol Intermediate (order based) 5-Amino-2,4,6- triiodisophthaloyl acid dichloride
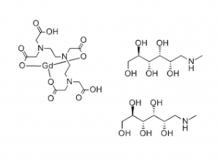
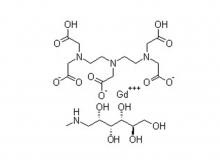
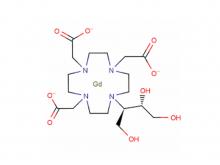
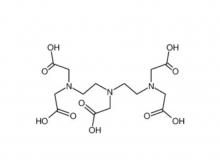 Diethylenetriaminepentaacetic acid (DTPA)
Diethylenetriaminepentaacetic acid (DTPA)

 EN
EN
 jp
jp  fr
fr  de
de  es
es  ru
ru  ar
ar 



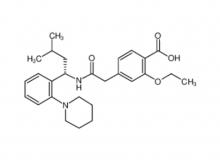
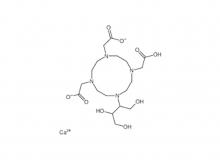









 Call us on:
Call us on:  Email Us:
Email Us:  No.3 Shuiyuan West Road, Miyun District, Beijing, China
No.3 Shuiyuan West Road, Miyun District, Beijing, China 